Customer Data Platform (CDP) for Pharma & Life Sciences across HCP and patient engagement
A Customer Data Platform (CDP) for Pharma & Life Sciences unifies the engagement data brand, medical, and field teams are permitted to use into a single profile per stakeholder, so engagement stays consistent across every touchpoint and stays inside the lines drawn by medical, legal, and regulatory review.

Unified HCP and patient engagement profiles
Bring HCP digital engagement, field rep call notes, content consumption, event participation, sample requests, and patient support programme interactions into one connected view per stakeholder.
Real-time engagement signals
Capture portal logins, content downloads, webinar attendance, RSVP responses, and patient app activity as they happen, so engagement reflects current interest rather than a quarterly extract.
Cross-system data unification
Connect Veeva CRM, content management systems, medical portals, event platforms, patient support programme tools, brand websites, and field force apps into one resolved record per HCP, patient, or KOL.
Engagement intelligence in context
Turn raw engagement data into clear signals about content affinity, channel preference, and topic interest that brand and field teams can act on within MLR-approved boundaries.
Trusted by thousands
Success Quantified
Real results from real customers—measured across ROI, growth and retention
500+
satisfied clients globally
98%
clients satisfaction
5x
ROI for clients
2x
more conversion
Core technology built for the regulated pharma stack
The infrastructure is shaped around how pharma data actually moves: high-frequency engagement data alongside slow-moving HCP master data, strict separation between commercial and medical activities, and the consent and compliance requirements that define what each role is allowed to see and act on.
Multi-identifier resolution across HCP and patient
Stitch identities across HCP master IDs (NPI, MCI, regional registry numbers), email, phone, portal accounts, and event registrations for HCPs, and across email, phone, app login, and programme enrolment ID for patients, with strict separation between the two domains.
Pharma-native data model
Pre-built schemas for HCPs, specialties, accounts, prescriptions (where permitted), field activities, samples, content engagement, events, and patient programme participation mean teams skip months of modelling and start activating data quickly.
Streaming profile updates
A medical portal login, a webinar registered, a content piece downloaded, a sample request submitted, a field rep call logged — every event flows into the profile within seconds and is available for triggers, segmentation, or rep lookups inside compliance boundaries.
How AI sharpens pharma engagement
Pharma engagement signals are sparse and regulated. An HCP might engage with a brand four times in a year across channels that are not connected, and the patterns that signal genuine interest, content fit, or rising influence are too subtle for rule-based logic. This is where models inside a Customer Data Platform (CDP) for Pharma & Life Sciences earn their place, applied within the boundaries set by medical, legal, and regulatory review.
Models score each HCP's likely interest in upcoming content, events, or rep visits based on past engagement, specialty, and channel preference. Field reps and brand teams reach out with content that fits the HCP rather than running a generic call cycle.
Models score each HCP's likely interest in upcoming content, events, or rep visits based on past engagement, specialty, and channel preference. Field reps and brand teams reach out with content that fits the HCP rather than running a generic call cycle.
For patients enrolled in support programmes, models score adherence risk based on app engagement, refill timing, support interactions, and self-reported milestones, so programme teams reach out with the right intervention before the patient lapses.
Models combine publication activity, congress participation, peer engagement, and digital influence signals to surface HCPs whose voice carries weight in their specialty, helping medical affairs teams prioritise scientific exchange and advisory engagement.
Use NVECTA Co-Pilot to ask things like "which oncologists in tier-1 cities have attended more than two of our webinars in the last six months and viewed the latest study summary" without writing SQL or waiting on the analytics team.
From unified profiles to pharma engagement impact
A live engagement view changes what brand, medical, and field teams can do day to day. The gains show up in HCP engagement quality, programme adherence, field force productivity, and the consistency of the brand experience across channels.
Hand field reps a clean view of each HCP's recent digital engagement, content interests, and unanswered medical information requests, so the next call builds on the last interaction instead of repeating it.
Reach patients with the right intervention — refill reminders, support content, counsellor outreach — based on adherence signals and engagement patterns rather than a fixed reminder schedule.
Match content drops, webinar invitations, and event RSVPs to the HCPs most likely to care, based on specialty, prior engagement, and topic affinity. Audiences receive what is genuinely relevant to their practice instead of every brand asset on the calendar.
Give brand managers, medical affairs, field force leadership, and patient programme teams role-appropriate access to the same engagement view, so the HCP experience and patient experience stay coherent across every team that touches them.
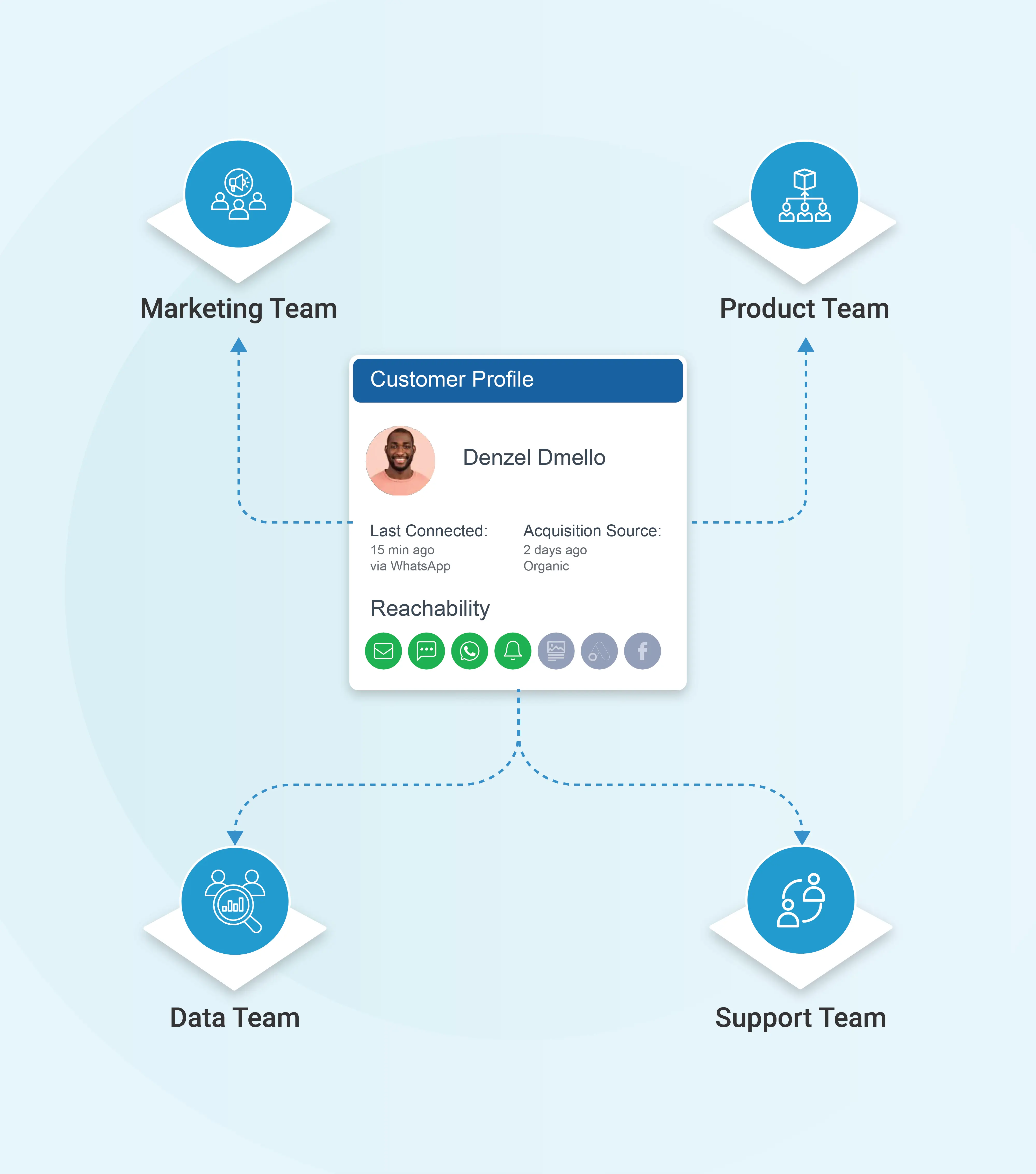
From unified profiles to pharma engagement impact
A live engagement view changes what brand, medical, and field teams can do day to day. The gains show up in HCP engagement quality, programme adherence, field force productivity, and the consistency of the brand experience across channels.
Hand field reps a clean view of each HCP's recent digital engagement, content interests, and unanswered medical information requests, so the next call builds on the last interaction instead of repeating it.
Reach patients with the right intervention — refill reminders, support content, counsellor outreach — based on adherence signals and engagement patterns rather than a fixed reminder schedule.
Match content drops, webinar invitations, and event RSVPs to the HCPs most likely to care, based on specialty, prior engagement, and topic affinity. Audiences receive what is genuinely relevant to their practice instead of every brand asset on the calendar.
Give brand managers, medical affairs, field force leadership, and patient programme teams role-appropriate access to the same engagement view, so the HCP experience and patient experience stay coherent across every team that touches them.

HCP, prescription, and field force data integration
Pharma data lives in some of the most fragmented and regulated systems in any industry. HCP master data sits in IQVIA OneKey or local equivalents. Prescription data flows through DDD or local syndicated feeds where compliant. Field activity sits in Veeva CRM. Content engagement is captured by approved content portals. Patient programme data lives in dedicated programme tools. The platform connects directly to Veeva CRM, IQVIA and local HCP master data sources, content portals, event platforms, medical information systems, and patient support programme tools, with strict role-based separation between commercial and medical data, and audit logs on every access.
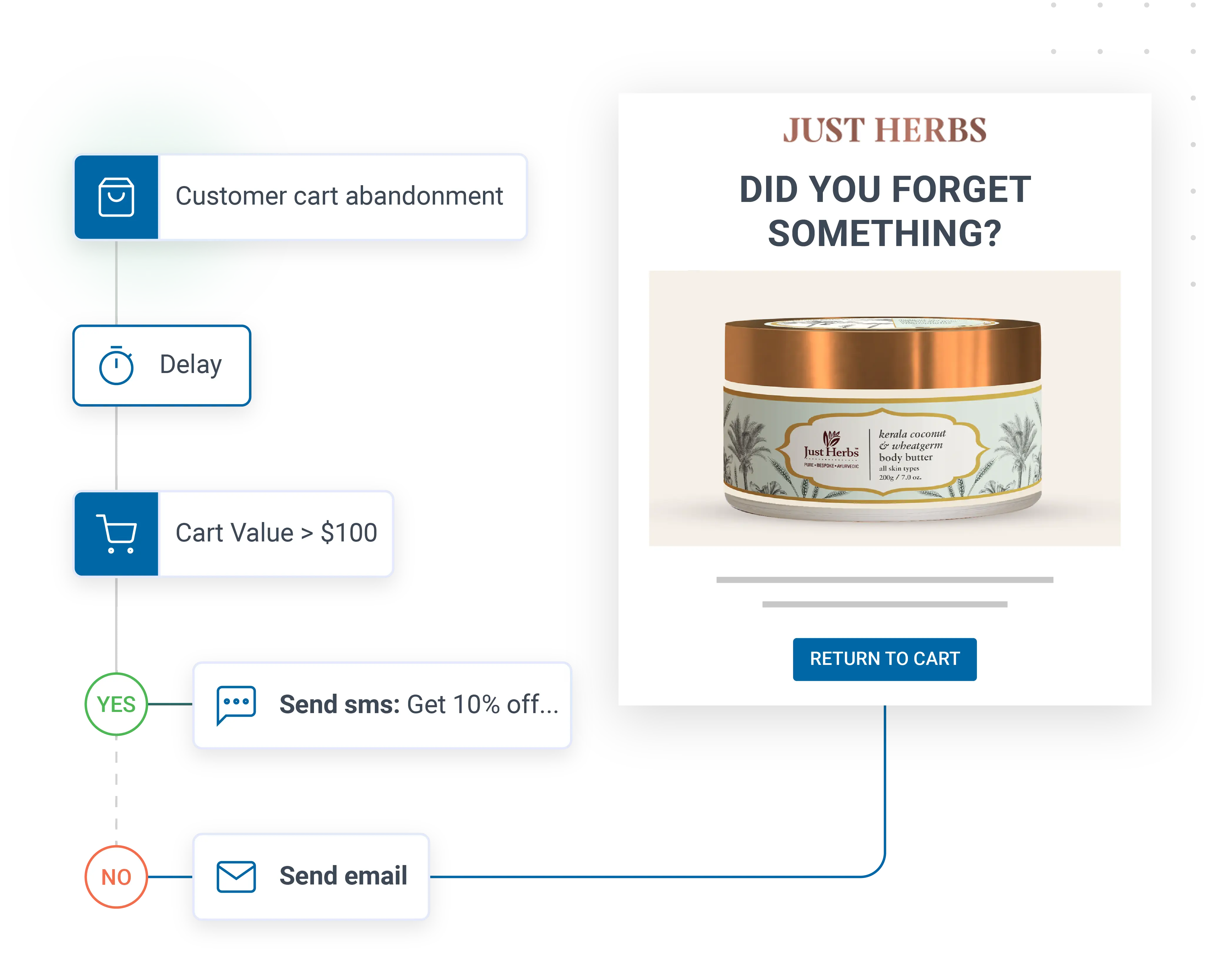
HCP, prescription, and field force data integration
Pharma data lives in some of the most fragmented and regulated systems in any industry. HCP master data sits in IQVIA OneKey or local equivalents. Prescription data flows through DDD or local syndicated feeds where compliant. Field activity sits in Veeva CRM. Content engagement is captured by approved content portals. Patient programme data lives in dedicated programme tools. The platform connects directly to Veeva CRM, IQVIA and local HCP master data sources, content portals, event platforms, medical information systems, and patient support programme tools, with strict role-based separation between commercial and medical data, and audit logs on every access.

HCP, patient, and field force engagement views
Pharma engagement typically involves four parties: the HCP who prescribes or recommends, the patient who takes the therapy, the field rep who carries the relationship, and the KOL who shapes scientific opinion. A Customer Data Platform (CDP) for Pharma & Life Sciences represents these relationships explicitly and separately. The HCP view holds engagement, content, and event history within MLR-approved boundaries. The patient view holds programme enrolment, adherence signals, and consented support interactions, kept fully separate from commercial data. The field rep view shows assigned HCPs, recent digital engagement, and suggested next actions. The KOL view tracks scientific engagement, publication activity, and advisory participation. Each view is governed by role-based access, regional regulations, and brand-specific MLR rules, so teams only see what they are allowed to see and act within the boundaries compliance has set.
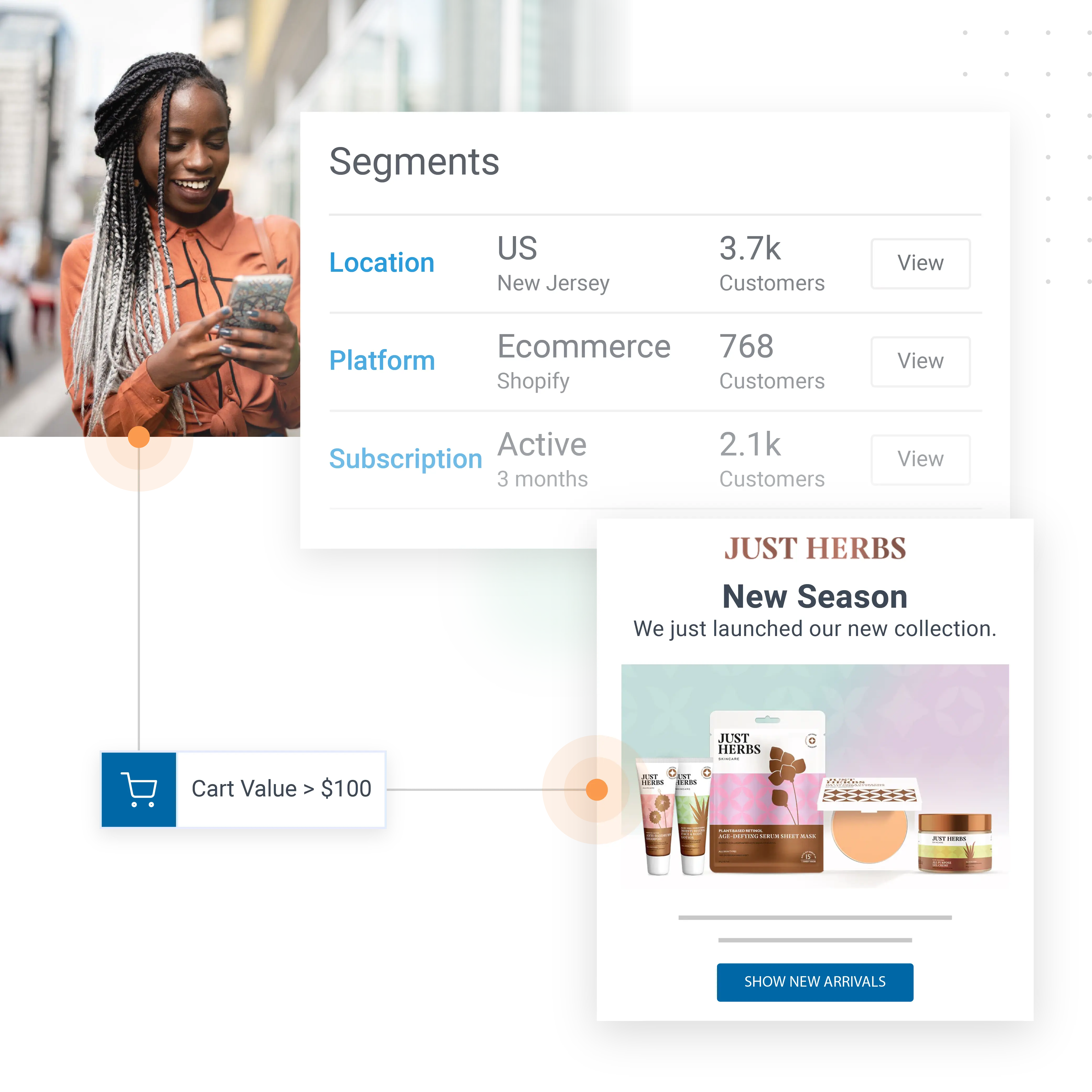

HCP, patient, and field force engagement views
Pharma engagement typically involves four parties: the HCP who prescribes or recommends, the patient who takes the therapy, the field rep who carries the relationship, and the KOL who shapes scientific opinion. A Customer Data Platform (CDP) for Pharma & Life Sciences represents these relationships explicitly and separately. The HCP view holds engagement, content, and event history within MLR-approved boundaries. The patient view holds programme enrolment, adherence signals, and consented support interactions, kept fully separate from commercial data. The field rep view shows assigned HCPs, recent digital engagement, and suggested next actions. The KOL view tracks scientific engagement, publication activity, and advisory participation. Each view is governed by role-based access, regional regulations, and brand-specific MLR rules, so teams only see what they are allowed to see and act within the boundaries compliance has set.
Your data remains in your control
Trusted by teams worldwide
NVECTA operates without duplicating or storing your data. Instead, we securely read directly from your existing data warehouse—so your data stays safe and untouched.



Seamless Integrations. Enterprise Ready
Connect your data, systems, and tools through 100+ reliable integrations, built to unify your stack and scale with your business.
FAQ section
It is a platform that unifies engagement data across HCPs, patients, and KOLs into single live profiles per stakeholder. It connects Veeva CRM, HCP master data, content portals, event platforms, patient programme tools, and digital channels so brand, medical, and field teams work from the same view within MLR-approved boundaries.
Veeva manages field force activities, call planning, and content delivery to HCPs. IQVIA provides syndicated HCP master data and, where compliant, prescription data. Neither unifies digital engagement, content consumption, event participation, and patient programme signals in real time across all channels. The CDP sits across these systems, resolves the engagement picture, and respects the strict commercial-medical separation pharma operates under.
The platform supports strict role-based separation between commercial and medical data, between HCP and patient domains, and between regions where regulations require it. Access is governed by role, brand, geography, and stakeholder type, with full audit trails on every access.
Yes. The data model handles prescription brands with HCP-led engagement, OTC brands with consumer-led engagement, animal health with veterinarian and pet-owner segments, and medical devices with HCP and institutional buyer segments, within the same platform.
Patient enrolment, consent, adherence signals, app engagement, support interactions, refill patterns, and self-reported outcomes are unified into a single patient profile within the programme. Adherence prediction, intervention triggers, and consented communication run on this view, kept fully separate from any HCP or commercial data.
Yes. Field rep calls, email engagement, webinar attendance, content portal activity, RSVP responses, sample requests, and medical information enquiries are all unified into the HCP profile, so omnichannel orchestration runs on real engagement signals rather than channel-specific guesses.
Built-in consent management aligned with HIPAA, GDPR, the Indian DPDP Act, regional pharma codes (PhRMA, EFPIA, IPA, ABPI), and adverse event reporting requirements. Every data access, every outbound communication, and every consent change is logged for audit. MLR review workflows can be integrated into content and engagement triggers where required.
Yes, where applicable. Patient health information is handled with the controls required under HIPAA in the US, GDPR Article 9 in the EU, and equivalent regional regulations, including encryption, access logging, data residency, and right-to-be-forgotten workflows.
Ready to transform your workflow?
Ready to unify your customer data and deliver personalized experiences?